|
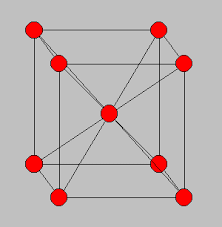
Every element has its own unique physical property. These properties are anything that has to do with their physical appearance, scent, texture, sometimes flavor, odor, size, things that it can do, and many other things. For appearance, francium is presumed to have a silvery, and gray color to it. No one knows the precise scent to it because it is very rare and usually found in uranium. The texture is metallic and also, it is presumed to be like that. No one has ever tasted francium because one, it would kill you, and two, it is a very unstable element. Again, scientist presume that it would be malleable because it is a metal and also, they presume that it is ductile. The ionic radius of a francium atom is 180 picometer. Francium`s predicted surface tension is 0.05092 Newts per meter at its melting point. It is predicted because it is insanely hard to actually have the time to figure it out. Because it is in the alkali metals, it is presumed to have similar properties, like being soft, malleable, ductile, and being good at conducting heat and electricity. Then of course, there is scent. Again, nobody was smell francium but it would probably not give out an odor because it is a metal. Finally, there is the density of francium. Density is how compact an element is. The more atoms it has in a set measurement, the denser it is. The density of Francium is 1.873 g/cm3 when at the temperature of 20 degrees Celsius. Last, the Structure of an Francium atom is in the form of a Body-Centered Cubic Bravais Crystal Lattice. This is how atoms are found and this structure is what they look like. It is also called a body centered cubic unit.
Background image provided by www.aiche.org/dippr
|
Photo produced by Pamela Smith
Produced by Alvesgaspar
Produced by Bart J. Zeghbroek
|